Goldsmiths, University of London (GUL) • Chemistry
Latest uploads for Chemistry at Goldsmiths, University of London (GUL). Looking for Chemistry notes at Goldsmiths, University of London (GUL)? We have lots of notes, study guides and revision notes available for Chemistry at Goldsmiths, University of London (GUL).
-
33
- 0
- 0
Modules Chemistry at Goldsmiths, University of London (GUL)
Notes available for the following courses of Chemistry at Goldsmiths, University of London (GUL)
Latest notes & summaries Goldsmiths, University of London (GUL) • Chemistry
a) Sulfur trioxide, SO3, is made industrially in enormous quantities by combining oxygen and sulfur dioxide, SO2. What amount (moles) of SO3 is represented by 2.60 kg of sulfur trioxide? b) Sulfur trioxide, SO3, is made industrially in enormous quantities by combining oxygen and sulfur dioxide, SO2. How many molecules are represented by 2.60 kg of sulfur trioxide? c) Sulfur trioxide, SO3, is made industrially in enormous quantities by combining oxygen and sulfur dioxide, SO2. How many sulf...
o Research the Emory University Planetarium and Observatory in Atlanta Georgia. Pretend you visited the planetarium or observatory in your area. o Write a 1,050- to 1,400-word report describing your experience: • The name of the facility and its association • The type of equipment • Showings (celestial objects observed) • Other things learned about astronomy • You can also include any planetarium show or videos you watched during your (mock) visit.
Biochemistry Assignment #3
Assignment: SCIE207 Phase 3 Lab Report Title: Lab to Determine the Outcome of Heredity Instructions: You will fill out the Punnett squares and answer a set of questions for each exercise.
HOMEWORK for Chapter 11: Gases
a) Calculate the total masses of the reactants in equation 4NH3(g) 5O2(g) → 4NO(g) 6H2O(g) Express your answer using four significant figures. b) Calculate the total masses of the products in equation above. Express your answer using four significant figures.
Question 1: a) Calculate the total masses of the reactants in equation 2SO2(g) O2(g) → 2SO3(g) Express your answer using five significant figures. b) Calculate the total masses of the products in equation above. Express your answer using four significant figures. Question 2: a) Calculate the total masses of the reactants in equation 4P(s) 5O2(g) → 2P2O5(s) Express your answer using four significant figures. b) Calculate the total masses of the ...
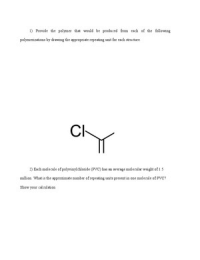
1) Provide the polymer that would be produced from each of the following polymerizations by drawing the appropriate repeating unit for each structure. 2) Each molecule of polyvinylchloride (PVC) has an average molecular weight of 1.5 million. What is the approximate number of repeating units present in one molecule of PVC? Show your calculation
Question 1: A tank of oxygen holds 28.0 L of oxygen (O2) at a pressure of 16.0 atm . When the gas is released, it provides 310. L of oxygen. What is the pressure of this same gas at a volume of 310. L and constant temperature? Express your answer with the appropriate units.