Chemistry
Popular textbooks · Show all (10) Hide

Cambridge International AS and A Level Chemistry Coursebook with CD-ROM
Lawrie Ryan, Roger Norris
34 documents
Adult Education
14 documents
All 6.833 results
Sort by

-
Test Bank for Organic Chemistry, 4th Edition by David R. Klein | 9781119659594|Chapter 1-27 | All Chapters with Answers and Rationals
- Exam (elaborations) • 2151 pages • 2023
- Available in package deal
-
- $24.14
- 15x sold
- + learn more
Test Bank for Organic Chemistry 4e 4th Edition by David R. Klein. ISBN-13: 9594 Full chapters test bank PDF TABLE OF CONTENTS 1 A Review of General Chemistry: Electrons, Bonds, and Molecular Properties 1 1.1 Introduction to Organic Chemistry 2 1.2 The Structural Theory of Matter 3 1.3 Electrons, Bonds, and Lewis Structures 4 1.4 Identifying Formal Charges 7 1.5 Induction and Polar Covalent Bonds 8 1.6 Reading Bond-Line Structures 11 1.7 Atomic Orbitals 14 1.8 Valence Bond Theory 17 1.9 Molecular...

-
Pearson Edexcel Level 3 GCE In Chemistry (9CH0) Advanced PAPER 1: Advanced Inorganic and Physical Chemistry QUESTION PAPER AND MARK SCHEME (MERGED)
- Exam (elaborations) • 76 pages • 2024
-
- $9.99
- + learn more
Pearson Edexcel Level 3 GCE In Chemistry (9CH0) Advanced PAPER 1: Advanced Inorganic and Physical Chemistry QUESTION PAPER AND MARK SCHEME (MERGED)

-
Pearson Edexcel Level 3 GCE In Chemistry (8CH0) Advanced Subsidiary PAPER 1: Core Inorganic and Physical Chemistry MERGED QUESTION PAPER AND MARK SCHEME
- Exam (elaborations) • 72 pages • 2024
-
- $9.99
- + learn more
Pearson Edexcel Level 3 GCE In Chemistry (8CH0) Advanced Subsidiary PAPER 1: Core Inorganic and Physical Chemistry MERGED QUESTION PAPER AND MARK SCHEME

-
AS-level AQA Chemistry questions and answers
- Exam (elaborations) • 16 pages • 2024
-
- $7.99
- + learn more
What is the charge of a proton? - ANSWERS +1 What is the charge of a neutron? - ANSWERS 0 What is the charge of an electron? - ANSWERS -1 What is the mass of a proton? - ANSWERS 1 What is the mass of a neutron? - ANSWERS 1 What is the mass of an electron? - ANSWERS 1/1800 What is an isotope? - ANSWERS Isotopes of an element are atoms with the same number of protons but different numbers of neutrons. Why do isotopes of an element all have the same chemical properties? - ANSWERS This is...
It is a well written notes of Haloalkanes and Haloarenes .I have written it with the help of NCERT book,and internet websites .These notes are best for board exams as well as for JEE and NEET.
Simplified note of periodic table in one go
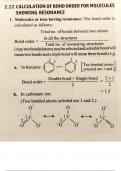
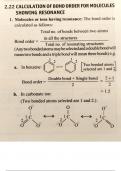
this document is about the bond order of molecules and formula to calculate bond order
this document is about the bond order of molecule and formula to calculate the bond order
these are the precised notes about the type of intramolecular forces in moleculaes and bond order of molecules , formula to calculate bond order for different molecules.

-
CHEM TEST 3 Question & Answers
- Exam (elaborations) • 7 pages • 2024
-
- $7.99
- + learn more
CHEM TEST 3 Question & Answers Anti-bonding orbitals * - ANSWER: atomic orbitals subtract (destructive interference) to form ____ these have 1 more node than the individual atomic orbitals, so they are higher in energy As the number of lone pairs increase around the central atom, the bond angle decreases. Why? - ANSWER: the increase in repulsive forces between lone pair-lone pair electron groups Axial position - ANSWER: above and below the plane which the central atom and equatorial bonds...
These are important formulas for calculating numericals in chemistry.