Ionic compunds Study guides, Class notes & Summaries
Looking for the best study guides, study notes and summaries about Ionic compunds? On this page you'll find 10 study documents about Ionic compunds.
All 10 results
Sort by

-
ACS Chemistry Final general Chem 1 Review 63 Questions with Verified Answers,100% CORRECT
- Exam (elaborations) • 7 pages • 2024
-
- $11.09
- + learn more
ACS Chemistry Final general Chem 1 Review 63 Questions with Verified Answers Density - CORRECT ANSWER mass / volume Conversion from celsius to kelvin - CORRECT ANSWER K = DegC + 273.15 Conversion from DegF to DegC - CORRECT ANSWER DegC = (5/9) (DegF - 32) Conversion from DegC to DegF - CORRECT ANSWER DegF = (9/5)(DegC) + 32 Intensive properties - CORRECT ANSWER Independent of amount of matter examined Extensive propertires - CORRECT ANSWER Dependent of amount of matter examined...

-
Chem 161 Final Exam Answers latest update 2023 rated A+
- Exam (elaborations) • 11 pages • 2023
-
- $9.99
- + learn more
Chem 161 Final Exam Answers latest update 2023 rated A+ Beta particle emission that is a high energy electron. the electron comes from a neutron so when it is emitted, the atomic number goes up by one, but the atomic mass remains the same alpha particle emission that is basically a helium nucleus (2+ charge), so when it is emitted the atomic number goes down by 2 and the atomic mass goes down by 4 Alkali metals first column Alkaline earth metals second column halogens group befor...

-
CLEP Biology: Chemistry of Biology Latest Review 2023/2024
- Exam (elaborations) • 6 pages • 2023
- Available in package deal
-
- $7.99
- + learn more
atoms -->The building blocks of all matter. properties of matter -->results of the structure of atoms and their interactions with each other. element -->A substance that can't be broken down into any other substance. The simplest form of an element is an atom. Protons -->Positive charged particles in the nucleus of an atom. Neutron -->Neutral charged particles in the nucleus of an atom. electrons -->Negative charged particles of an atom. They orbit the nucleus. They have mu...

-
CLEP Biology Exam Latest Updates
- Exam (elaborations) • 18 pages • 2023
- Available in package deal
-
- $11.99
- + learn more
CLEP Biology Exam Latest Updates atoms - The building blocks of all matter. properties of matter - results of the structure of atoms and their interactions with each other. element - A substance that can't be broken down into any other substance. The simplest form of an element is an atom. Protons - Positive charged particles in the nucleus of an atom. Neutron - Neutral charged particles in the nucleus of an atom. electrons - Negative charged particles of an atom. They orbit t...

-
CHEM 120 FINAL EXAM REVIEW | CHEM 120 FINAL EXAM practice problems
- Exam (elaborations) • 11 pages • 2022
-
- $8.49
- + learn more
2. Which of the following elements a) is a main group element: Mn, Ag, Co, F, La ______________ b) exists as a diatomic molecule: He, N, Al, P, Hg ______________ c) is a transition element: B, S, Cr, Ne, Sr _____________ d) is a noble gas: H, Be, F, Ar, Au ___________ e) is a metalloid: O, C, Ti, Ge, Cl ___________ f) most likely to gain an electron when forming an ion Na, Mg, Al, Br, He ______________ 3. Indicate the charges of the following ions: a) an ion formed from Ba: ______ b)...

-
Final Exam Practice Questions Chemistry 103- General Chemistry 2023
- Exam (elaborations) • 6 pages • 2023
-
- $9.99
- + learn more
Which nitrogen - containing compound is one cause of acid rain? - ANS-NO2 How many moles of gas would be found in a rigid 5.0L cylinder at 12.0 atm pressure and 15 degrees C - ANS-2.5 mol Which is the IUPAC name for Cu2SO3? - ANS-copper (I) sulfite NH3, H2S, NO2, and Cl2 each has a distinct odor. If you were standing one meter away when bottles of each of these gases were opened simultaneously, which gas would you smell first? - ANS-NH3 Which statement best describes the change phase o...

-
Chapter 10 ~ Properties of Solutions
- Class notes • 1 pages • 2023
-
- $7.99
- + learn more
This chapter covers solubility and solutions as a whole. It discusses electrolytes and how it relates to electricity, there are many examples covering how to understand solubility. As well as how to relate solubility to ions, acids, bases, etc.

-
Chapter 10 ~ Properties of Solutions
- Class notes • 1 pages • 2023
-
- $7.99
- + learn more
This chapter covers solubility and solutions as a whole. It discusses electrolytes and how it relates to electricity, there are many examples covering how to understand solubility. As well as how to relate solubility to ions, acids, bases, etc.

-
Chapter 10 ~ Properties of Solutions
- Class notes • 1 pages • 2023
-
- $7.99
- + learn more
This chapter covers solubility and solutions as a whole. It discusses electrolytes and how it relates to electricity, there are many examples covering how to understand solubility. As well as how to relate solubility to ions, acids, bases, etc.
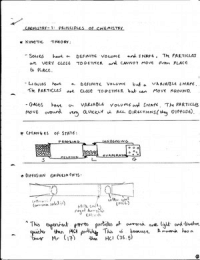
-
IGCSE Chemistry- Principles of chemistry
- Summary • 10 pages • 2015
-
- $4.55
- + learn more
- States of matter - Atoms - Atomic structure - Relative formula masses and molar volumes of gases - Chemical formulae and chemical equations - Ionic compunds - Covalent substances - Metallic substances - Electrolysis



