P1
Sodium Carbonate Titration
Titration is a volumetric procedure, involving many diferent apparatuses to
make it work. The accuracy of the results depend on the concentration of the
regent used to do the titration.
Apparatus
Goggles
100cm3 Beaker
250cm3 conical fask
25cm3 Pipettes
25cm3 Burette
Clamp stand and holder
Plastic flter funnel
Phenolphthalein indicator solution
Standard sodium carbonate solution
1 M hydrochloric acid
Pipette fller
Method
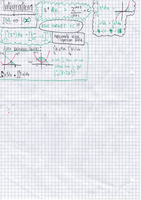
1. Transfer a 25cm3 aliquot (portion) of your sodium carbonate solution to a
250cm3 capacity conical fask..
2. A pipette is used to measure accurately a volume of this solution - for
example, 10cm3. A safety pipette fller is used to draw solution into the
pipette. This is emptied into a conical fask. A few drops of Phenolphthalein
indicator solution may be added to the conical fask. This will show a
change of colour when the titration is complete.
3. A second chemical is placed in a burette. This other solution is of a
chemical that will react with the synthesised chemical sample in the
conical fask. Often the solution in the burette is an acid or alkali, and it
must be of a precise, known concentration.
4. The solution from the burette is run into the conical fask. The solution is
added one drop at a time, with swirling to mix the solutions as the end-
point is approached. Eventually, a colour change shows that the correct
amount has been added to react completely with the synthesised
chemical in the sample.
5. The end-point of the titration is when the solution just changes from pink
to colourless. Repeat 1-4 thrice to get three separate results. The results
need to reliable which is the reason the test is repeated
Photos
1
BTEC LEVEL 3 APPLIED SCIENCE EXTENDED DIPLOMA
Unit 4: Scientifc Practical Techniques
Sodium Carbonate Titration
Titration is a volumetric procedure, involving many diferent apparatuses to
make it work. The accuracy of the results depend on the concentration of the
regent used to do the titration.
Apparatus
Goggles
100cm3 Beaker
250cm3 conical fask
25cm3 Pipettes
25cm3 Burette
Clamp stand and holder
Plastic flter funnel
Phenolphthalein indicator solution
Standard sodium carbonate solution
1 M hydrochloric acid
Pipette fller
Method
1. Transfer a 25cm3 aliquot (portion) of your sodium carbonate solution to a
250cm3 capacity conical fask..
2. A pipette is used to measure accurately a volume of this solution - for
example, 10cm3. A safety pipette fller is used to draw solution into the
pipette. This is emptied into a conical fask. A few drops of Phenolphthalein
indicator solution may be added to the conical fask. This will show a
change of colour when the titration is complete.
3. A second chemical is placed in a burette. This other solution is of a
chemical that will react with the synthesised chemical sample in the
conical fask. Often the solution in the burette is an acid or alkali, and it
must be of a precise, known concentration.
4. The solution from the burette is run into the conical fask. The solution is
added one drop at a time, with swirling to mix the solutions as the end-
point is approached. Eventually, a colour change shows that the correct
amount has been added to react completely with the synthesised
chemical in the sample.
5. The end-point of the titration is when the solution just changes from pink
to colourless. Repeat 1-4 thrice to get three separate results. The results
need to reliable which is the reason the test is repeated
Photos
1
BTEC LEVEL 3 APPLIED SCIENCE EXTENDED DIPLOMA
Unit 4: Scientifc Practical Techniques