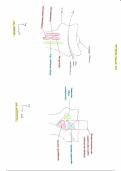
Half-Reaction E° (V)
Li+ + e- -> Li -3.04
K+ + e- -> K -2.92
Ca2+ + 2e- -> Ca -2.87
Na+ + e- -> Na -2.71
Mg2+ + 2e- -> Mg -2.37
Al3+ + 3e- -> Al -1.66
Zn2+ + 2e- -> Zn -0.76
Fe2+ + 2e- -> Fe -0.44
Ni2+ + 2e- -> Ni -0.25
Pb2+ + 2e- -> Pb -0.13
H+ + e- -> 1/2 H2 0.00
Cu2+ + 2e- -> Cu +0.34
Ag+ + e- -> Ag +0.80
Hg2 2+ + 2e- -> 2Hg +0.85
Fe3+ + e- -> Fe2+ +0.77
Cl2 + 2e- -> 2Cl- +1.36
MnO4- + 8H+ + 5e- -> Mn2+ + 4H2O +1.51
O2 + 4H+ + 4e- -> 2H2O +1.23
F2 + 2e- -> 2F- +2.87