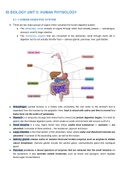
CFCs
CFCs (chlorofluorocarbons) are used as:
Refrigerants
Propellants for aerosols
Generating foamed plastics like expanded polystyrene or polyurethane foam
Solvents for dry cleaning
General degreasing purposes1
If ozone levels are reduced more harmful uv radiation reaches the Earth's surface and can lead to medical
problems such as increased risk of sunburn and skin cancer and it also accelerates skin aging processes. The
formation of chlorine atom radicals is the root of the problem because they readily react with ozone and change it
back to much more stable ordinary oxygen.
CFCs diffuse up into the stratosphere and decompose when hit by ultraviolet light to produce free radicals,
including free chlorine atoms.2
Sweden was the first country to ban CFC containing aerosol sprays in 1978. This was followed by an
international agreement called the 1987 “Montreal Protocol”, aiming to limit use of ozone-destroying
substances. A production end date was given for CFC-containing solvents, foaming agents and aerosol
sprays, and in 1990, an objective was set to reduce CFCs by 50% by 1999. 3
O + O2 ⇌O3
Ozone formation and destruction in the stratosphere is an example of a dynamic equilibrium.4
Photodissociation of CF2Cl2 in UV:
CF2Cl2 -> CF2Cl. + Cl.
Ozone depletion due to Cl radicals
Propagation 1 Cl. + O3 -> ClO. + O2
Propagation 2 ClO. + O -> Cl. + O2
Ozone depletion due to Nitrogen oxide radicals
Propagation 1 NO. + O3 -> NO2. +O2
Propagation 2 NO2. + O -> NO. + O2
The overall reaction for both examples is O3+ O ->2O2 5
1
https://www.chemguide.co.uk/organicprops/haloalkanes/uses.html
2
https://docbrown.info/page07/ASA2group7f.htm#:~:text=The%20chemically%20very%20stable%20CFCs,are%20highly%20reactive%20free%20radicals.
3
https://www.researchgate.net/publication/260266128_Chlorofluorocarbons_CFCs_and_their_effect_on_the_atmosphere
4
http://butane.chem.uiuc.edu/pshapley/genchem2/a9/book.pdf
5
A Level Chemistry A for OCR Student Book
CFCs (chlorofluorocarbons) are used as:
Refrigerants
Propellants for aerosols
Generating foamed plastics like expanded polystyrene or polyurethane foam
Solvents for dry cleaning
General degreasing purposes1
If ozone levels are reduced more harmful uv radiation reaches the Earth's surface and can lead to medical
problems such as increased risk of sunburn and skin cancer and it also accelerates skin aging processes. The
formation of chlorine atom radicals is the root of the problem because they readily react with ozone and change it
back to much more stable ordinary oxygen.
CFCs diffuse up into the stratosphere and decompose when hit by ultraviolet light to produce free radicals,
including free chlorine atoms.2
Sweden was the first country to ban CFC containing aerosol sprays in 1978. This was followed by an
international agreement called the 1987 “Montreal Protocol”, aiming to limit use of ozone-destroying
substances. A production end date was given for CFC-containing solvents, foaming agents and aerosol
sprays, and in 1990, an objective was set to reduce CFCs by 50% by 1999. 3
O + O2 ⇌O3
Ozone formation and destruction in the stratosphere is an example of a dynamic equilibrium.4
Photodissociation of CF2Cl2 in UV:
CF2Cl2 -> CF2Cl. + Cl.
Ozone depletion due to Cl radicals
Propagation 1 Cl. + O3 -> ClO. + O2
Propagation 2 ClO. + O -> Cl. + O2
Ozone depletion due to Nitrogen oxide radicals
Propagation 1 NO. + O3 -> NO2. +O2
Propagation 2 NO2. + O -> NO. + O2
The overall reaction for both examples is O3+ O ->2O2 5
1
https://www.chemguide.co.uk/organicprops/haloalkanes/uses.html
2
https://docbrown.info/page07/ASA2group7f.htm#:~:text=The%20chemically%20very%20stable%20CFCs,are%20highly%20reactive%20free%20radicals.
3
https://www.researchgate.net/publication/260266128_Chlorofluorocarbons_CFCs_and_their_effect_on_the_atmosphere
4
http://butane.chem.uiuc.edu/pshapley/genchem2/a9/book.pdf
5
A Level Chemistry A for OCR Student Book