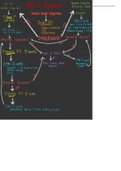
Charge on an electron is 1.602x10-19C NA = 6 1023 mol-1
Mass of an electron is 9.1x10-31 kg exp = 2.72
Planck’s constant has a numerical value of 6.626x10-34 J s
The Boltzmann Constant has a value of 1.38 x 10 -23 J K-1
Speed of light c= 299,792,458 m/s
I. Converting the units of energy/wavelength
E=vh= hc/=hcṽ
V*e=1eV= 1.6e-19 J
[v]=Hz=s-1
[ṽ]=cm-1
Test problems:
a) Spectrometer detected light at 500 nm (electronic spectra). Determine the wavenumber of
light as well at its frequency and energy of the photon. (20000 cm-1 and 3.98e-19 J)
b) Spectrometer detected light at 1700 cm-1 (vibrational). Determine the wavelength of light (in
nm) as well at its frequency and energy of the photon. (6m or 6e-6 m and 3.38e-20)
c) Spectrometer detected light at 10 cm-1 (rotational). Determine the wavelength of light as
well at its frequency and energy of the photon. (1e6 nm or1e-3m and 1.99e-22 J)
d) The energy of the molecular bond is 3eV. Light of what colour would you use to break the
bond?
II. Boltzman distribution
Population of levels
Test problems:
a) Calculate the relative population of first excited vs ground state levels for electronic (500
nm), vibrational (1700 cm-1) and rotational (10 cm-1) transitions at room temperature? Use
the energies calculated above
N1/No=2.24e-43 (500 nm); 2.4e-4 (1700 cm-1) and 0.95 (10 cm-1)
b) How would these distributions change at 1000 K
III. Absorption
Beer-Lambert Law
Mass of an electron is 9.1x10-31 kg exp = 2.72
Planck’s constant has a numerical value of 6.626x10-34 J s
The Boltzmann Constant has a value of 1.38 x 10 -23 J K-1
Speed of light c= 299,792,458 m/s
I. Converting the units of energy/wavelength
E=vh= hc/=hcṽ
V*e=1eV= 1.6e-19 J
[v]=Hz=s-1
[ṽ]=cm-1
Test problems:
a) Spectrometer detected light at 500 nm (electronic spectra). Determine the wavenumber of
light as well at its frequency and energy of the photon. (20000 cm-1 and 3.98e-19 J)
b) Spectrometer detected light at 1700 cm-1 (vibrational). Determine the wavelength of light (in
nm) as well at its frequency and energy of the photon. (6m or 6e-6 m and 3.38e-20)
c) Spectrometer detected light at 10 cm-1 (rotational). Determine the wavelength of light as
well at its frequency and energy of the photon. (1e6 nm or1e-3m and 1.99e-22 J)
d) The energy of the molecular bond is 3eV. Light of what colour would you use to break the
bond?
II. Boltzman distribution
Population of levels
Test problems:
a) Calculate the relative population of first excited vs ground state levels for electronic (500
nm), vibrational (1700 cm-1) and rotational (10 cm-1) transitions at room temperature? Use
the energies calculated above
N1/No=2.24e-43 (500 nm); 2.4e-4 (1700 cm-1) and 0.95 (10 cm-1)
b) How would these distributions change at 1000 K
III. Absorption
Beer-Lambert Law