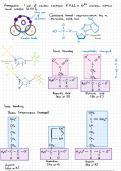
Arogadro I mol :
of carbon contains 6 022x10
. carbon atoms
and weighs 12 011 .
.
q
Lone
pair
of Covalent bond :
represented by a
·
electrons straight , solid line HzN
·
B
O N
- N
* H HO P O
O
N
N
Glycosidia
Covalent bonds O M bond
OH
0-
P
lonic bonding negatively charged
CH3
-
O O
0-
.n
O
Phosphodiester
io
41
H H
0- g
- &
H
31 C
band
i
O
ciz
O O C
~
P
CH2 I
-
-
O O
O N N
CH2
CH2
-
N
N
+
HaNt-c- C - o I
III HzNt 0
H
H -
"H H H
-
C -
C -
n
↓
-
HO
Ho
O
Aspartic Acid Glutamic acid
(Asp or D) (Gl orE)
lonic bonding
Basic (positively Charged NH2
NHzt
I
!
c =
NH2t
CH2 NH
+
I NH I
CH2
I I CH2
I
CH2
I
NH
CH
CH2 CH2
H2
HzNt -
C
I
-
C
II
- o HyNt-c
I
-
C
II
-
O
Hant -
k -c
Il
-
o
HO H O H O
Histidine Arginine
(His H) (Ara R)
usine
or or
k)
, Van der Waals Hydrogen bonding
S-
Weak interactions between
O
-
two dipolar atoms .
Hydrogen - -.
-
STEP I
-
-
band
↑~ H H
11ll St St
- t - x - t -
O
- -
H H
STEP 2
-
-
-
t
- -
---- - -
-
-
Hydrogen bonding in biomolecules : Interaction between an electropositive H
(0 or N)
in one molecule and an
electronegative atom in another molecule·
Bond DNA .
that joins base pairs in
H
St 5
0 - - - - -
H - N
---
N
-
-
o 6 St
-
+
Y N
N - H - ---- N
N
"N
s
5 -
N St
- - - - -
H -
- N g-
- Y R N H O R
- - - -
-
N
-
I
O I H
Adenine Thymine Guanine Cytosine
Gdst :
↳highlyParamagneticexcellentMRcontrastae t
↳ can replace calcium in the
body
-
O
9 o
/
vi
- O2H