PCM III Final Exam (Based on Exam Breakdown PPT)2022
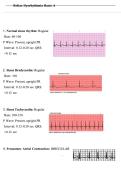
L MCA Ischemic Stroke Pathophysiology -Frontal: impulse, planning movement (apraxia) -Pre-Central Gyrus: Motor control -Post-Central Gyrus: sensory reception -Parietal lobe: orientation in space, lateropulsion (pusher's), limb position during mvmt -Temporal: sound, long term memory -Basal Ganglia: motor planning -Broca's: expressive aphasia -Wernicke's: receptive aphasia L MCA Ischemic Stroke Sensory & Motor Impairments -CNs - corticobulbar tract -Unilateral tongue innervation -Sparing of mm of mastication and ability to wrinkle forehead due to B innervation (CN VII) -CN XI - low incidence of SCM impairment after stroke L MCA Ischemic Stroke Mobility & Functional Limitations ROM, MMT, sensation, pain, spasticity, balance, bed mobility, gait, endurance, functional status, ADLs, task analysis L MCA Ischemic Stroke Aerobic Exercise -Aerobic exercise 1) 20 - 60 min/ session or Multiple 10 min bouts 2)40 - 70% HRR 3) 3 - 5/wk L MCA Ischemic Stroke OMs -Borg RPE -Berg Balance -Stroke Rehab Assessment of Mvmt (STREAM) Upper Limb Subscale -TUG BORG RPE -6 (no exertion) -11 (light) -13 (somewhat hard) -20 (maximal exertion) berg balance scale interpretation - 41-56 = low fall risk - 21-40 = medium fall risk - 0-20 = high fall risk *a change of 8 points required to reveal a genuine change in fxn btwn 2 assessments Chronic Kidney Disease (CKD) pathophysiology -common causes: diabetes, HTN, glomerulonephritis ESRD (end stage renal disease) -final phase of kidney disease -loss of kidney fxn -requires dialysis/kidney transplant -uremia cluster -reduced glomerular filtration rate (15 mL/min; 90 mL/min = normal) -Increased phosphate levels -Decreased Ca levels stimulus to parathyroid gland increased PTH release increased Ca uptake in kidneys (BUT kidneys are not fxning) -Cardiovascular: CAD, HTN, CHF, pulmonary edema, dyspnea, pericarditis -Leading cause of death: chest p!, nausea, SOB, sweating MSK issues assoc w/ ESRD -renal osteodystrophy 1) renal rickets: bone weakness & deformation 2) osteomalacia: demineralization of bone 3) osteitis fibrosis: inflammation of bone & fibers that connect to bone -bone pain: worse w/ exercise & WB -fx: spine & long bones CKD & ESRD endurance, mobility & functional limitations -Sensory/motor neuropathy -pain -SOB -decreased functional capacity (60 - 70% of norm) -HR is unreliable indicator of exertion -decreased endurance and functional status CKD & ESRD Interventions -Use Borg RPE to monitor exercise tolerance -Dialysis- recommend routine exercise. Avoid exercises the day before dialysis. -Exercise testing not recommended b/c mm fatigue limits testing procedure. 4 - 6/wk -Low - Mod intensity, 2-3 sets, 8 - 15 reps *Take BP in UE opposite AV shunt CKD & ESRD OMs -Borg RPE -Berg -TUG R-Sided HF Pathophysiology -raised pulmonary artery pressure -backs up in R atrium & venous vasculature -raised jugular venous distention -peripheral edema R-Sided HF & Dependent Edema OMs -6 MWT -Modified Borg Dyspnoea Scale -10 M Walk Test -Dyspnea Management Questionnaire COPD Pathophysiology -chronic airway inflammation & remodeling -characterized by: airway narrowing, parenchymal destruction, pulmonary vascular thickening -risk factors: 1) cigarette smoking #1, 2) organic/inorganic dusts, inhaling toxins 3) pollutants (outdoor pollution, indoor 2nd hand smoke) 4) genetic alpha 1 antitrypsin deficiency and lack of surfactant 5) host factors (hyperreactivity of airways, overall lung growth) -airway damage leads to airway repair/remodeling; common in small airways (bronchioles) -hypersecretion of goblet cells -decreased ciliary fxn (air enters, trapped in distal airways) chronic bronchitis -inflammation of lining of bronchial tubes, daily cough/mucus production -Blue bloaters, mucus obstructs gas exchange/rattles, prolonged expiratory wheezing/crackles on ausculatation/ decreased breath sounds -Chronic productive cough for 3mo for 2 consecutive yrs -X-ray: cardiac silhouette gets small as heart is pushed by hyperinflated lungs, excess body fluids, SOB emphysema -alveoli destroyed from cigarette smoke and other irritating gas/particulate matter -Pink puffers, enlargement of air space distal to terminal bronchiole -X-ray: hyperinflated lungs (dark/black) centrilobar emphysema loss of respiratory bronchioles in prox portion of acinus w/ sparing of distal alveoli) panlobar emphysema permanent destruction of entire acinus distal to respiratory bronchioles w/ no obvious associated fibrosis Distal acinar emphysema localized around septae of lungs, can lead to pneumothorax COPD Gold Classification FEV/FVC 70% -I Mild: w/ or w/out cough & sputum; FEV1 ≥80% -II Mod: SOB w/ exertion, w/ or w/out cough sputum; FEV1 50 - 79% -III Severe: greater SOB w/ exercise, decreased capacity/ fatigue/ repeated exacerbation; FEV1 30-49% -IV Very Severe: chronic respiratory failure; FEV1 30% manisfestations of COPD -Dyspnea: during exertion & lower level activity -Enlarged thorax: barrel shape, loss of lung elastic recoil -Hyperinflation, cough, ventilation perfusion mismatching -Hypoxemia, hypercapnia, clubbing -Polycythemia- enrythropoietin stimulated b/c of low O2 levels COPD tests & measures -Chest x-rays: low flat diaphragm, horizontal ribs, enlarged R ventricle, barrel shape chest, elevated shoulder girdle -Auscultation: expiratory wheeze, crackles COPD interventions -O2 sats 88% Concerning, want to maintain @ least 90% O2 Sat -Non Pharm: O2 therapy (partial pressure 55 mmHg), arterial blood gas studies, exercise, pursed lip breathing -Pharm: antitussives (decrease cough), decongestants, mucolytic agents (break sulfide bonds of mucus, leukotrienes, anti-inflammatory, steroids, antibiotics CHF risk factors -HTN: 140/90... leads to L ventricular stiffening -CAD: fatty deposits, plaques, atherosclerosis -cardiomyopathy (fam hx), -valvular disease (congenital disease, heart mm pumps harder), -DM: pts develop hypolipidemia & HTN -aging (wt gain, increased adipose): impaired endothelium dependent vasodilation L ventricular stiffness poor diastole fxn ventricular arterial stiffening vascular dysfxn impaired regulation -hypercholesterolemia (HTN CAD), -hyperthyroidism -Elevated pulse pressure: good predictor of HF (every 10 mm increases risk of HF by 14%)!!!!!!!! HF syndrome -inability of weakened heart to maintain cardiac output, -impaired cardiac pump fxn, -blood backs up into venous/pulmonary circulation --congestion of blood into body tissue & lungs -high pressure in pulmonary capillaries results in pulmonary HTN systolic failure -The left ventricle loses its ability to contract normally -reduces SV/CO/EF -HF w/ reduced ejection fraction diastolic failure -L ventricle stiff & unable to relax -reduced SV/CO -EF is unaltered -HF w/ preserved ejection fraction -Damage to heart mm reduces CO triggers renin angiotensin aldosterone sys facilitates Na and H2O retention in renal tubules increased blood vol, cardiac tissue undergoes remodeling (hypertrophy) functional classification of CHF -I: absence of dyspnea w/ daily activity (w/out limitation) -II: ordinary activity can trigger fatigue (slight limitation) -III: less than ordinary activity can cause fatigue, palpations, dyspnea (marked limitation) -IV: s/s of HF evident @ rest (discomfort w/ all physical activity) manifestations of CHF -Dyspnea: worse when lying down or w/ increased activity and PND -Fatigue: heart can't adequately pump blood and O2 -Cough: caused by backing up of fluid into longs (blood tinged sputum) -EF: 55% - 70% (systolic 40%) CHF tests & measures -Percussion, Locomotion, Balance, Mobility, Auscultation, Heart sounds/adventitious sounds, ROM, MMT, -Diagnostic tools: ECG (abnormal if EF is reduced), chest x-rays (cardiomegaly, opacities from pulmonary edema, blunting costophrenic angle), echocardiography (wall thickness, chamber size, LV fxn), lab findings (elevated BNP, normal levels 100pg/mL, 400 pg/mL indicator of HF) normal values CVP 0-8 *raised in RV failure or biventricular failure normal values PAP 9-19 *raised in LV failure PCWP 6-12 *raised in LV failure & biventricular failure BNP released by cardiac cells when endothelial cells are stretched *strong predictor of poor prognosis of HF preserved EF = lower BNP levels CHF interventions -Non-Pharm: 1) O2 therapy 2) heart healthy diet (mediterranean diet) 3) exercise -Surgical: 1)LVAD 2)heart transplant 3)ICD 4)pacemaker -Pharm: 1)alpha/beta blockers 2)ACE inhibitors (block conversion of angiotensin I to II, limiting vasoconstriction) 3) ARB (prevent angiotensin II form binding to receptors limiting vasocontriction) 4) vasodilators 5) positive inotropes (digoxin), 6)mineralocorticoid receptor antagonists 7)diuretics (decrease workload by decreasing total vol) -Incentive spirometry criteria to terminate/modify exercise in CHF patient -RR40 bpm -S3 heart sounds -pulmonary crackles -increase in CVP by 10 mmH L side HF pathophysiology -reduced CO -blood backs up in L atrium & lungs -SOB -cough orthopnea, cardiomegaly, L-sided HF test & measures -heart sounds -lung auscultation DM Type II pathophysiology -Cellular level, cells not sensitive to insulin -Chronic, systemic disorder results in excess glucose in blood (hyperglycemia) and disruption of normal metabolism. Results in defect in action of insulin -Later, gradual onset (40yo) -90% of cases -Obesity-associated resistance -Need hypoglycemic agents, exercise, diet, and wt control fasting plasma glucose values -norm: 100 mg/dl -prediabetes: 100-125 mg/dl -diabetes: 125 mg/dl *higher risk for diabetes & heart disease if pt has prediabetes, HTN, and dyslipidemia DM Type II risk factors -fam hx -ethnicity (blk) -BMI 25 -sedentary -gestational diabetes -vascular disease -HTN -HDL 35 -triglycerides 250 -smoking DM Type II complications -heart & kidney disease -blindness -limb amputation DM Type II manifestations -Polyuria/polydipsia/ blurred vision/ dizziness/ weakness/fatigue -Atherosclerosis -Retinopathy -Nephropathy (Leading cause of ESRD) -Infection (reduced wound healing) -MSK probs -Sensory, motor, autonomic neuropathies (deformities, ulcerations) -Cognitive decline (executive fxn declines) DM Type II integ, sensory, motor impairments -Infection- decreased wound healing -Ulcerations -MSK probs- UE manifestations, arthritis, spine, osteoporosis -Sensory, motor, autonomic neuropathies -Cognitive decline -Balance DM Type II tests & measures -Post-sx status (CV and respiratory) -ROM -MMT -reflexes -sensation -functional status (bed mobility, transfers, gait) -amputated extremity (type of post-sx dressing) -intact limb (ABI, pedal pulses, skin integrity, wounds) DM Type II interventions -Goal is for pt is to drop 5-10% body wt and 150 min of mod exercise/wk -30-60 min of mod aerobic exercise 5x/wk w/ 3-5/wk resistance exercises and flexibility exercises; fruit as a snack every 30 mins and pasta/crackers after -Safe level: 100-200 mg/dl "safe" (≤ 100, give a carb snack and wait 15 min and retest) -2 cups of water, exercise w/in 2hrs of last meal -No injection in mm being used w/ resistance day of exercise -Protect limb, independence w/ mobility, proper positioning/WB precautions, education, independent inspection of B feet, full ROM of BLE, return to strength, independent w/ all functional mobility, improved strength, low risk for fall, K-level 2 or 3 DM Type II pre-prosthetic interventions -CV using UE bike -positioning (hip flexor stretch), elevate above heart to reduce swelling -balance: standing -mobility: gait training -residual limb care: inspect for odor/drainage -Pt ed DM Type II post-prosthetic interventions -higher level gait training -progress AD DM Type II intervention contraindications -poor control of glucose -poorly controlled retinopathy/HTN/neuropathy/nephropathy/dehydration -extreme temps DM Type II OMs -tinetti -bates-Jensen wound assessment tool -diabetes foot screen CRPS Type II Pathophysiology -clear hx of nerve trauma/injury -triggers: diabetic neuropathy, amputation, fx/sprain/soft tissue injury/sx/needle stick -abnormal response that magnifies effects of injury/excessive response to a trigger -peripheral abnormalities -maladaptive CNS reorganization -causalgia -neuropathic pain -neuropathy CRPS Type II peripheral abnormalities -axons/Type C fibers, carry pain messages to BVs, molecules secreted from terminals of hyperactive nerve fibers contribute to inflammation & BV abnormalities 1) dilated BV: leak fluid into interstitial tissue, cause red/swollen skin 2) constricted BVs: decreased O2 & nutrients, cause cold/white/blue skin; underlying mm/tissues starved of O2, cause mm/jt pain 3) BV dilation/constriction controlled by small nerve fibers & chemical messengers in blood CRPS Type II maladaptive CNS reorganization occurs quickly w/ prolonged pain/immobilization CRPS Type II causalgia -primary lesion, peripheral -burning pain related to diabetes, pain after an amputation CRPS Type II neuropathic pain caused by lesion/disease of somatosensory nervous system CRPS Type II neuropathy disturbance of fxn or pathological change in a nerve CRPS Type II tests & measures -Observe motor tasks and assess: symmetry, compensations, aberrant mvmts (scapulohumeral, lumbopelvic, painful arc, loss of prox control) -Assess cognitive-emotional factors: fear avoidance, pain catastrophizing, central sensitization -R/O arthritis, lyme disease, mm disease, small fiber neuropathies
Written for
- Institution
- USA PCM3
- Module
- USA PCM3
Document information
- Uploaded on
- October 31, 2022
- Number of pages
- 17
- Written in
- 2022/2023
- Type
- Exam (elaborations)
- Contains
- Questions & answers
Subjects
-
pcm iii final exam based on exam breakdown ppt2022
Also available in package deal