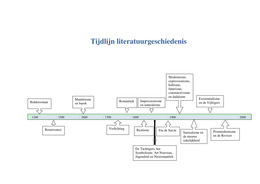
Atoms and reactions
The atom
JJ Thompson came up with the idea of the plum pudding model where an atom was
a positively charged sphere with negative electrons stuck in it.
Rutherford showed that the plum pudding model was wrong by alpha particles
(positively charged) at a thin sheet of gold foil. From the plum pudding model they
expected that most alpha particles would be deflected slightly but they found that
most alpha particles passed straight through. A small amount were deflected
backwards which disproved the plum pudding model. So Rutherford came up with
the nuclear model of the atom.
The Bohr model was a further improvement suggesting that electrons are fixed in
orbitals or shells which have fixed energies. When an electron moves between
shells, electromagnetic radiation of a fixed frequency is emitted or absorbed.
Relative mass
Relative atomic mass = the weighted mean mass of an atom of an element
compared with 1/12 of an atom of carbon-12.
Relative isotopic mass = the mass of an atom of an isotope compared with 1/12 of
the mass of an atom of carbon-12.
Relative molecular mass = the weighted mean mass of a molecule compared with
1/12 of an atom of carbon-12.
Relative formula mass = the weighted mean mass of a formula unit.
Isotope = an atom of an element with a different number of neutrons.
Calculating relative atomic mass from isotopic abundances:
Multiply each relative isotopic mass by its percentage relative isotopic
abundance and add up the results
Divide by 100
Mass spectrometry can be used to find percentage abundances of isotopes of an
element to work out the relative atomic mass of an element.
, Moles
A mole is the amount of any substance that has the same number of atoms as there
are carbon atoms in 12g of carbon-12.
The amount of substance is based on the standard count of atoms called Avogadro’s
constant – 6.02 x 1023.
Moles = number of atoms/Avogadro’s constant
Molar mass = total mass of an element, molecule or ion. Measured as mass per
mole of substance.
Avogadro’s law = under the same conditions of temperature and pressure, a mole of
gas will fill the same volume of space (24dm3 or 24000cm3) – known as molar gas
volume.
The ideal gas equation lets you find the number of moles in a certain volume at any
temperature or pressure – pV = nRT
Empirical formula = gives the smallest whole number ratio of atoms of each
element in a compound.
Find moles of each element
Divide moles of each by the smallest number of moles
Multiply ratio if need be to make whole numbers
Molecular formula = gives the actual number of atoms of each type of element in a
molecule.
Moles = mass/RFM OR mass = moles x RFM OR RFM = mass/moles
Water of crystallisation = water present in the crystalline structure of a compound
Calculate molar mass of anhydrous salt
Calculate moles of anhydrous salt
Calculate moles of water
Divide by smallest number of moles to find the ratio
Salts are ionic compounds; all solid salts consist of a lattice of positive and negative
ions and in some salts, water molecules are incorporated into the lattice too.
Acids and bases
Acids are proton donors. When mixed with water all acids dissociate to release H+
ions. Bases on the other hand are proton acceptors. Bases that are soluble in water
are known as alkalis and release OH- ions.
The atom
JJ Thompson came up with the idea of the plum pudding model where an atom was
a positively charged sphere with negative electrons stuck in it.
Rutherford showed that the plum pudding model was wrong by alpha particles
(positively charged) at a thin sheet of gold foil. From the plum pudding model they
expected that most alpha particles would be deflected slightly but they found that
most alpha particles passed straight through. A small amount were deflected
backwards which disproved the plum pudding model. So Rutherford came up with
the nuclear model of the atom.
The Bohr model was a further improvement suggesting that electrons are fixed in
orbitals or shells which have fixed energies. When an electron moves between
shells, electromagnetic radiation of a fixed frequency is emitted or absorbed.
Relative mass
Relative atomic mass = the weighted mean mass of an atom of an element
compared with 1/12 of an atom of carbon-12.
Relative isotopic mass = the mass of an atom of an isotope compared with 1/12 of
the mass of an atom of carbon-12.
Relative molecular mass = the weighted mean mass of a molecule compared with
1/12 of an atom of carbon-12.
Relative formula mass = the weighted mean mass of a formula unit.
Isotope = an atom of an element with a different number of neutrons.
Calculating relative atomic mass from isotopic abundances:
Multiply each relative isotopic mass by its percentage relative isotopic
abundance and add up the results
Divide by 100
Mass spectrometry can be used to find percentage abundances of isotopes of an
element to work out the relative atomic mass of an element.
, Moles
A mole is the amount of any substance that has the same number of atoms as there
are carbon atoms in 12g of carbon-12.
The amount of substance is based on the standard count of atoms called Avogadro’s
constant – 6.02 x 1023.
Moles = number of atoms/Avogadro’s constant
Molar mass = total mass of an element, molecule or ion. Measured as mass per
mole of substance.
Avogadro’s law = under the same conditions of temperature and pressure, a mole of
gas will fill the same volume of space (24dm3 or 24000cm3) – known as molar gas
volume.
The ideal gas equation lets you find the number of moles in a certain volume at any
temperature or pressure – pV = nRT
Empirical formula = gives the smallest whole number ratio of atoms of each
element in a compound.
Find moles of each element
Divide moles of each by the smallest number of moles
Multiply ratio if need be to make whole numbers
Molecular formula = gives the actual number of atoms of each type of element in a
molecule.
Moles = mass/RFM OR mass = moles x RFM OR RFM = mass/moles
Water of crystallisation = water present in the crystalline structure of a compound
Calculate molar mass of anhydrous salt
Calculate moles of anhydrous salt
Calculate moles of water
Divide by smallest number of moles to find the ratio
Salts are ionic compounds; all solid salts consist of a lattice of positive and negative
ions and in some salts, water molecules are incorporated into the lattice too.
Acids and bases
Acids are proton donors. When mixed with water all acids dissociate to release H+
ions. Bases on the other hand are proton acceptors. Bases that are soluble in water
are known as alkalis and release OH- ions.