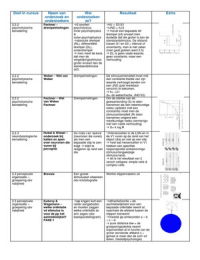
1 Polarity 5 TemperatureControl
HighSpecificHeat H2Oresists
unequalsharing ofelectrons make water changes in temperature dueto
a polarmoleculeWater ispolarbecausethe the presence ofhydrogen bonds
oxygenendhasa partial negativechargeand Heatis absorbedto break
thehydrogen endhasapartialpositivecharge hydrogenbonds heat is released
coughnat
whenhydrogen bondsform
moderates airtemp
largebodiesof waterabsorb heat
hygiene duringthedayandrelease heata
night stabilizes oceantempt
organisms resist changes in
2 Cohesion internal temperature be mostcells
attraction ofmoleculesforothermolecules aremade of water
ofthesamekind watermolecules are Evaporativecooling waterhasahigh
attracted tothemselves heatotevaporation
hydrogenbondsbetweenH2omolecules hold moderates Earth'sclimate
themtogether andincrease cohesive prevents overheatingsweating
allowsforthetransportofwaternutrients
against gravity in plants 6 Density
responsibleforsurfacetension aswatersolidities it expands and
measureofhowdifficultit is to stretch becomeslessdense
or break thesurfaceofa liquid Hydrogenbondscausewater
3Adhesion molecules toformacrystalline
structure oice.islessdense
clingingofone moleculeto a different
molecule
duetopolarity of 20
7 Solvent
thedissolving agentin a solution
4 Capillary Action watercan dissolvealmostanything
upward movement of waterupa narrow w a charge
tube againsttheforce ofgravity due to waterformsH bondswiththe sugar
cohesion adhesion andsurfacetension orprotein todissolve it
occurswhen adhesion isgreater than
cohesion
HighSpecificHeat H2Oresists
unequalsharing ofelectrons make water changes in temperature dueto
a polarmoleculeWater ispolarbecausethe the presence ofhydrogen bonds
oxygenendhasa partial negativechargeand Heatis absorbedto break
thehydrogen endhasapartialpositivecharge hydrogenbonds heat is released
coughnat
whenhydrogen bondsform
moderates airtemp
largebodiesof waterabsorb heat
hygiene duringthedayandrelease heata
night stabilizes oceantempt
organisms resist changes in
2 Cohesion internal temperature be mostcells
attraction ofmoleculesforothermolecules aremade of water
ofthesamekind watermolecules are Evaporativecooling waterhasahigh
attracted tothemselves heatotevaporation
hydrogenbondsbetweenH2omolecules hold moderates Earth'sclimate
themtogether andincrease cohesive prevents overheatingsweating
allowsforthetransportofwaternutrients
against gravity in plants 6 Density
responsibleforsurfacetension aswatersolidities it expands and
measureofhowdifficultit is to stretch becomeslessdense
or break thesurfaceofa liquid Hydrogenbondscausewater
3Adhesion molecules toformacrystalline
structure oice.islessdense
clingingofone moleculeto a different
molecule
duetopolarity of 20
7 Solvent
thedissolving agentin a solution
4 Capillary Action watercan dissolvealmostanything
upward movement of waterupa narrow w a charge
tube againsttheforce ofgravity due to waterformsH bondswiththe sugar
cohesion adhesion andsurfacetension orprotein todissolve it
occurswhen adhesion isgreater than
cohesion