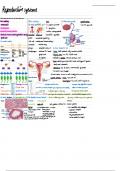
Topic 12: Alkenes
Structure, bonding and reactivity
Alkenes are unsaturated hydrocarbons meaning they contain a carbon-carbon double bond.
This is an area of high electron density making it susceptible to attack from electrophiles
(species that are attracted to ∂- areas). It consists of a normal covalent bond and a π bond.
Bromine water is used to identify this double bond and other unsaturated compounds. It
turns the solution from orange-brown to colourless if a double bond is present in the
substance.
Structure, bonding and reactivity
Alkenes are unsaturated hydrocarbons meaning they contain a carbon-carbon double bond.
This is an area of high electron density making it susceptible to attack from electrophiles
(species that are attracted to ∂- areas). It consists of a normal covalent bond and a π bond.
Bromine water is used to identify this double bond and other unsaturated compounds. It
turns the solution from orange-brown to colourless if a double bond is present in the
substance.