pH curves
HELLO
Buffer solutions
Resist changes in pH after adding small amounts of acid/base
Acidic buffers
Weak acid and salt of that acid
E.g. ethanoic acid and sodium ethanoate
- If H+ ions are added
- They combine with ethanoate ions to form ethanoic acid
- If OH- ions are added
- It reacts with the acid to form water
How acidic buffers are made
- Strong base and excess weak acid
Alkali buffers
Weak base with salt of that base
E.g. ammonia and ammonium chloride
- If H+ ions are added
- They combine with OH- to form water
- Ammonia dissociates to replace ions
- If OH- ions are added
- It reacts with ammonium ions to ammonia
Salt hydrolysis
- Neutral salts- strong acids with strong base
- Alkaline salts- weak acid with strong base
- Acidic salts- strong acid with weak base
, Acid-base titrations
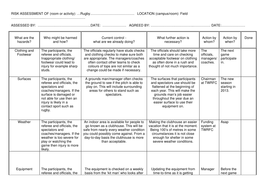
Strong acid with strong base
E.g. HCl + NaOH
- 50 cm3 1.0M of each
1. Only HCl in conical flask
- pH 0
2. Adding 49cm3 NaOH
- pH 2
3. Adding all NaOH
- pH 7
Equivalence (infection) point - pH 7
Weak acid with strong base
E.g. CH3COOH + NaOH
- 50 cm3 1.0M of each
Weak acid will have a buffer effect
- Initially resist change
- Inflexion point higher pH
pH values calculated with Ka
Indicator: Phenolphthalein (9.6)
HELLO
Buffer solutions
Resist changes in pH after adding small amounts of acid/base
Acidic buffers
Weak acid and salt of that acid
E.g. ethanoic acid and sodium ethanoate
- If H+ ions are added
- They combine with ethanoate ions to form ethanoic acid
- If OH- ions are added
- It reacts with the acid to form water
How acidic buffers are made
- Strong base and excess weak acid
Alkali buffers
Weak base with salt of that base
E.g. ammonia and ammonium chloride
- If H+ ions are added
- They combine with OH- to form water
- Ammonia dissociates to replace ions
- If OH- ions are added
- It reacts with ammonium ions to ammonia
Salt hydrolysis
- Neutral salts- strong acids with strong base
- Alkaline salts- weak acid with strong base
- Acidic salts- strong acid with weak base
, Acid-base titrations
Strong acid with strong base
E.g. HCl + NaOH
- 50 cm3 1.0M of each
1. Only HCl in conical flask
- pH 0
2. Adding 49cm3 NaOH
- pH 2
3. Adding all NaOH
- pH 7
Equivalence (infection) point - pH 7
Weak acid with strong base
E.g. CH3COOH + NaOH
- 50 cm3 1.0M of each
Weak acid will have a buffer effect
- Initially resist change
- Inflexion point higher pH
pH values calculated with Ka
Indicator: Phenolphthalein (9.6)