keywords key phrases
C1 Atomic Structure
C1.1 - Atoms
❖ There are about 100 atoms naturally found on Earth and everything is made of atoms as
they are the smallest known unit
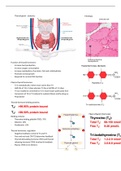
➢ atoms are made up of a tiny centre-of-mass, the nucleus (which contains protons
and neutrons) as well as being orbited by shells of electrons
❖ Certain substances are made up of only one type of atom - these are elements
➢ since there are only roughly 100 different atoms, then that must mean that there
are only roughly 100 different types of elements
➢ like how atoms have different structures, different elements also have different
properties
■ there are metal and non-metal classifications, so for example, metals like
gold, copper and silver are lustrous and malleable but non-metals like
oxygen, chlorine and nitrogen are gases at room temperature
❖ Certain substances are made up of different types of atoms - these are compounds
➢ chemical bonds between atoms hold them tightly together in a compound
❖ scientists use the Periodic Table to list all the chemical elements, split into eight groups
which are elements with similar chemical properties
From:
https://www.google.com/url?sa=i&url=https%3A%2F%2Fwww.sciencenewsforstudents.org%2Farticle%2Fscientists-say-periodic-table&psig=A
OvVaw0hOfPzVkh__jgdXmTB4MNj&ust=1653340868212000&source=images&cd=vfe&ved=0CAwQjRxqFwoTCIDm5uuE9PcCFQAAAAAdA
AAAABAE
❖ each column in the Periodic Table is a group
C1.2 - Chemical Equations
, keywords key phrases
❖ Chemical equations show the reactants (the starting substances) and the products
(the new substances made)
➢ for example, water is made like this:
❖ To find out how much of each substance takes part in the reaction, you would
have to look at a symbol equation
❖ On each side of the equation, the amount of type of atom should be the same, as
the Law of Conservation of Mass states: “the total mass of products formed in a
reaction is equal to the total mass of the reactants”
➢ state symbols (s, l, g, aq) can also be added to produce information about
the states of the the products and reactants
C1.3 - Separating Mixtures
Compounds: two or more elements bonded together in a fixed ratio
Mixtures: two or more substances (elements or compounds) that are not chemically
combined, and so retain their individual properties
Compounds Mixtures
- fixed ratio of elements - proportions vary based on how
- chemical reactions must be used to much of each substance is mixed
separate elements in the together
compound - physical means (filtration,
- chemical bonds between atoms crystallisation, distillation,
chromatography) can be used to
C1 Atomic Structure
C1.1 - Atoms
❖ There are about 100 atoms naturally found on Earth and everything is made of atoms as
they are the smallest known unit
➢ atoms are made up of a tiny centre-of-mass, the nucleus (which contains protons
and neutrons) as well as being orbited by shells of electrons
❖ Certain substances are made up of only one type of atom - these are elements
➢ since there are only roughly 100 different atoms, then that must mean that there
are only roughly 100 different types of elements
➢ like how atoms have different structures, different elements also have different
properties
■ there are metal and non-metal classifications, so for example, metals like
gold, copper and silver are lustrous and malleable but non-metals like
oxygen, chlorine and nitrogen are gases at room temperature
❖ Certain substances are made up of different types of atoms - these are compounds
➢ chemical bonds between atoms hold them tightly together in a compound
❖ scientists use the Periodic Table to list all the chemical elements, split into eight groups
which are elements with similar chemical properties
From:
https://www.google.com/url?sa=i&url=https%3A%2F%2Fwww.sciencenewsforstudents.org%2Farticle%2Fscientists-say-periodic-table&psig=A
OvVaw0hOfPzVkh__jgdXmTB4MNj&ust=1653340868212000&source=images&cd=vfe&ved=0CAwQjRxqFwoTCIDm5uuE9PcCFQAAAAAdA
AAAABAE
❖ each column in the Periodic Table is a group
C1.2 - Chemical Equations
, keywords key phrases
❖ Chemical equations show the reactants (the starting substances) and the products
(the new substances made)
➢ for example, water is made like this:
❖ To find out how much of each substance takes part in the reaction, you would
have to look at a symbol equation
❖ On each side of the equation, the amount of type of atom should be the same, as
the Law of Conservation of Mass states: “the total mass of products formed in a
reaction is equal to the total mass of the reactants”
➢ state symbols (s, l, g, aq) can also be added to produce information about
the states of the the products and reactants
C1.3 - Separating Mixtures
Compounds: two or more elements bonded together in a fixed ratio
Mixtures: two or more substances (elements or compounds) that are not chemically
combined, and so retain their individual properties
Compounds Mixtures
- fixed ratio of elements - proportions vary based on how
- chemical reactions must be used to much of each substance is mixed
separate elements in the together
compound - physical means (filtration,
- chemical bonds between atoms crystallisation, distillation,
chromatography) can be used to