4.3 Particle model of matter
4.3.1 Changes of state and the particle model
4.3.1.1 Density of materials
Density is the amount of mass in a given volume. It tells us how tightly matter is
packed together
- Depends on what it is made of
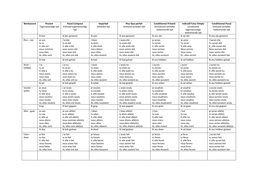
To find the density of an object, you can fill a water jug and record how much there is
to start with. Then you can place the object inside it and see how much water it
displaced – volume of the object
- Or you can use a displacement can and fill it with water until it starts coming out.
Then you can place a measuring cylinder beneath the spout. Then you can place
the object in and see how much water is filled in the cylinder, indicating the
volume of the object. Then you should measure the mass and use the density
equation to find the density
The density of a material is defined by:
- Density = mass/volume
- Kg/m^3 = kg / m^3
- Volume is a measure of how much space something takes up. 1ml=1cm^3
The particle model can be used to explain:
- The different states of matter
- Differences in density
- The forces between them can be seen
4.3.1.2 Changes of state
When substances change state (melt, freeze, boil, evaporate, condense or sublimate),
mass is conserved
Changes of state are physical changes which differ from chemical changes because
the material recovers its original properties if the change is reversed. They don’t form
a new substance. The number of particles doesn’t change – they are just arranged
differently – this means mass is conserved
, Solid:
- Strong forces of attraction hold the particles close together in a fixed, regular
arrangement
- The particles don’t have much energy so they can only vibrate about their fixed
positions
- The density is generally highest in this state as the particles are closest together
Liquid:
- There are weaker forces of attraction between the particles
- The particles are close together, but can move past each other and form irregular
arrangements
- For any given substance, in the liquid state its particles will have more energy than
in the solid state
- They move in random directions at low speeds
- Liquids are generally less dense than solids
Gas:
- There are almost no forces of attraction between the particles.
- For any given substance, in the gas state its particles will have more energy than in
the solid or liquid state
- They are free to move and travel in random directions at high speeds
- They have low densities
Melting point is the temperature when liquids turn to solid and solids turn to liquid
Boiling point is the temperature when gas turns to liquid or liquids turn to gas
Difference between evaporation and boiling
Evaporation happens at a liquids surface when the liquid is below its boiling point – it
can happen at any temperature below boiling point
At its boiling point, a liquid boils because bubbles of gas are formed inside the liquid –
it only happens at a substances boiling point
4.3.2 Internal energy and energy transfers
4.3.2.1 Internal energy