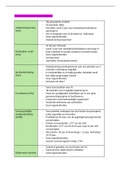
Periodic Table and Energy
PERIODICITY : 3.1.1
Periodic trend in electron configuration across period 2 and 3
Period 2 has only two outer shells
Period 3 has three outer shells
Period 3 has larger atomic radius therefore it is less reactive as it will
require more energy to gain an electron as the electrostatic forces of
attraction is weaker
where is the s block
Group 1 and Group 2
Including helium
where is the p block
Group 3 , 4 , 5 , 6 , 7 and 0
where is the d block
Transition metals
what is first ionisation energy
Amount of energy required to remove 1 mol of electrons from 1 mole of
gaseous atoms to form one mole of 1+ ions
what is the trend of first ionisation energy across periods
Across a period the shielding remains the same so it doesn't effect it
The nuclear charge increases as the proton number increases
Therefore there is stronger electrostatic forces of attraction across a period
Stronger electrostatic forces means smaller atomic radius as you go across
as outer electrons are more attracted to the nucleus and pulled in closer
Therefore the ionisation energy increases as you go across the period
Periodic Table and Energy 1
, what is the trend of first ionisation energy down groups
As you go down the shielding increases as there are more electron shells
The atomic radius increases and nuclear charge increases
Ionisation energy decreases as you go down the group
why is there a change in trend from O to N
O has a pair of opposite spin electrons in the first p orbital that will repel
each other and thus require less energy so the first ionisation enrgy would
be lower than N instead of higher
N electron configuration = 1s2 2s2 2p3
O electron configuration = 1s2 2s2 2p4
why is there a change in trend from B to Be
Ionisation energy in boron is lower
Boron has an unpaired electron in one 2p orbital which is a higher energy
level
Easier to remove electron from higher energy level when the elctron is
unpaired therefore less energy is required
Be electron configuration = 1s2 2s2
B electron configuration = 1s2 2s2 2p1
what is second ionisation energy
energy required to remove one mole of electrons from one mole of 1+
gaseous ions to form one mole of 2+ gaseous ions
what is the trend of melting and boiling point across period 3
the melting point increases from Group 1 to Group 3 because they are
metallic lattices
Decrease from group 4 to group 5 due to change from giant covalent
structure (Si) to simple covalent molecule (P)
(P) forms P4 molecules which is very small so the london forces are very
weak so less energy needed than Sulphur
Periodic Table and Energy 2
PERIODICITY : 3.1.1
Periodic trend in electron configuration across period 2 and 3
Period 2 has only two outer shells
Period 3 has three outer shells
Period 3 has larger atomic radius therefore it is less reactive as it will
require more energy to gain an electron as the electrostatic forces of
attraction is weaker
where is the s block
Group 1 and Group 2
Including helium
where is the p block
Group 3 , 4 , 5 , 6 , 7 and 0
where is the d block
Transition metals
what is first ionisation energy
Amount of energy required to remove 1 mol of electrons from 1 mole of
gaseous atoms to form one mole of 1+ ions
what is the trend of first ionisation energy across periods
Across a period the shielding remains the same so it doesn't effect it
The nuclear charge increases as the proton number increases
Therefore there is stronger electrostatic forces of attraction across a period
Stronger electrostatic forces means smaller atomic radius as you go across
as outer electrons are more attracted to the nucleus and pulled in closer
Therefore the ionisation energy increases as you go across the period
Periodic Table and Energy 1
, what is the trend of first ionisation energy down groups
As you go down the shielding increases as there are more electron shells
The atomic radius increases and nuclear charge increases
Ionisation energy decreases as you go down the group
why is there a change in trend from O to N
O has a pair of opposite spin electrons in the first p orbital that will repel
each other and thus require less energy so the first ionisation enrgy would
be lower than N instead of higher
N electron configuration = 1s2 2s2 2p3
O electron configuration = 1s2 2s2 2p4
why is there a change in trend from B to Be
Ionisation energy in boron is lower
Boron has an unpaired electron in one 2p orbital which is a higher energy
level
Easier to remove electron from higher energy level when the elctron is
unpaired therefore less energy is required
Be electron configuration = 1s2 2s2
B electron configuration = 1s2 2s2 2p1
what is second ionisation energy
energy required to remove one mole of electrons from one mole of 1+
gaseous ions to form one mole of 2+ gaseous ions
what is the trend of melting and boiling point across period 3
the melting point increases from Group 1 to Group 3 because they are
metallic lattices
Decrease from group 4 to group 5 due to change from giant covalent
structure (Si) to simple covalent molecule (P)
(P) forms P4 molecules which is very small so the london forces are very
weak so less energy needed than Sulphur
Periodic Table and Energy 2