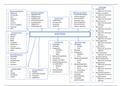
1.1 Fundamental particles
The subatomic particles
Atoms are made up of three fundamental particles: protons, neutrons and
electrons.
The protons and neutrons form the nucleus in the centre of the atom.
● Protons and neutrons can also be called NUCLEONS because they are
located in the nucleus of the atom.
● The electrons surround and orbit the nucleus in energy levels/shells.
Proton Neutron Electron
Relative MASS 1 1 1/1840
Relative CHARGE +1 0 -1
The arrangement of subatomic particles
● Protons and neutrons are located in the atom’s
nucleus. They are held together by a force called
the strong nuclear force.
● Electrons surround the nucleus in energy
levels/shells and orbit the nucleus.
● The electrostatic forces of attraction are the forces that hold the
electrons and protons together in the atom.
● The strong nuclear force is MUCH stronger than the electrostatic forces
of attraction, so it overcomes the repulsion between the protons in the
nucleus. It only acts over short distances within the nucleus.
, Atoms and ions
Ions form when an atom loses or gains electron(s) to fill its outer shell. They do
this in order to become more stable.
Metal atoms: Non-metal atoms:
Metal atoms lose electrons to form Non-metal atoms gain electrons to
positive ions. These ions have more form negative ions. These ions
positive protons than negative contain more negative electrons than
electrons. positive protons
Differences between atoms and ions:
- Atoms have an overall neutral charge, whereas ions have a negative or
positive charge.
- Atoms have an EQUAL number of protons and electrons, but ions have
different numbers of protons and electrons.
How to work out the number of electrons in an ION:
- Positive ion: atomic number - charge
- Negative ion: atomic number + charge
Electron arrangement in atoms
Energy levels:
● Electrons in different shells have different amounts of ENERGY, and so
can be represented on an energy level diagram.
● The number of electrons in each energy level diagram is given by the
formula 2(n2), where n is the number of the shell.
Shell: MAXIMUM NO. OF ELECTRONS:
,1 2 x (12) = 2
2 2 x (22) = 8
3 2 x (32) = 18
4 2 x (42)= 32
Shells and subshells:
● Shells are the MAIN energy levels (1,2,3 etc.)
● Shells are then split into SUBLEVELS → s, p, d, f
Shell: Maximum no. of Subshells:
electrons:
1 2 s
2 8 s, p
3 18 s, p, d
4 32 s, p, d, f
, Orbitals
● Subshells are split into ORBITALS
● Each orbital can hold a maximum of two electrons with OPPOSITE spins.
SHELL: MAX NO. OF ELECTRONS: SUBSHELLS: ORBITALS:
1 2 s One s orbital
2 8 s,p One s orbital
Three p orbitals
(8 electrons: 2 are
in one s orbital. 6
electrons remain
so they are
arranged in three
p orbitals
because orbitals
hold a maximum
of two electrons
only!)
3 18 s, p, d One s orbital
Three p orbitals
Five d orbitals
4 32 s, p, d, f One s orbital
Three p orbitals
Five d orbitals
Seven f orbitals
Atomic orbitals