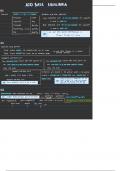
IB CHEMISTRY:
TOPIC 9 REDOX PROCESSES NOTES
9.1 Oxidation and reduction
Oxidation and Reduction
o In oxidation and reduction reactions (also called redox reactions), electrons move
between atoms
o Oxidation involves the loss of Oxidation Reduction
electrons. The more a substance is
oxidized, the more positive it gets Loss of electrons Gain of electrons
o Reduction involves the gain of
electrons. The more a substance is Loss of hydrogen Gain of hydrogen
reduced the more negative it gets
o Remember: OIL RIG (Oxidation is Gain of hydrogen Loss of oxygen
loss, reduction is gain)
Oxidant and reductant
Definitions
Oxidant – A substance that readily oxidizes other substances
Reductant – A substance that readily reduces other substances
• An oxidizing agent makes oxidation
Oxidizing Reducing
happen. In order to make oxidation
agent/oxidant agent/reductant
happen, the agent must take the electron
from the substance
Gains electrons Loses electrons
• So, oxidizing agents are reduced
• Common oxidizing agents include Oxidizes another Reduces another reactant
halogens, ozone, maganate (VII) ions and reactant
hydrogen peroxide
• A reducing agent makes reduction Is reduced during the Is oxidized during the
happen. In order to make reduction reaction reaction
happen, the agent must give an electron
to the substance
• So, reducing agents are oxidized
• Common reducing agents include hydrogen, carbon, carbon monoxide and sulfur dioxide
, Oxidation state
• The oxidation state (also known as oxidation number) is the total number of electrons
that an atom either gains or loses to form a chemical bond with another atom – (the
charge an atom would have if the compound was composed of ions)
• Oxidation numbers are represented by a roman number. Example: Cu2O: Copper (I)
oxide, FeCl2: Iron (II) chloride
• Oxidation states can be used to identify which species have been oxidized and which
have been reduced
o An increase in oxidation number means oxidation (so reducing agent) (Loss
of electrons, becomes more positive)
o A decrease in oxidation number means reduction (so oxidizing agent) (Gain
of electrons, becomes more negative)
• Oxidation states are written as NUMBER and then SIGN
• To assign oxidation states to atoms in a molecule or compound we follow some very
specific rules:
Sum of oxidation state for a neutral Sum of oxidation state for a polyatomic ion: Ion
compound: 0 charge
Element by itself: 0 Group 1/2/3: Always +1/+2/+3
Monatomic ion: Ion charge D-Block elements have variable oxidation states
Halogens: Usually -1 (+1 with oxygen) Hydrogen: +1 (-1 with metals)
Oxygen: -2 (-1 in peroxide) Florine: Always -1
Question: Deduce the oxidation state of Cr in K2Cr2O7
𝐾: +1 (2 × +1) = +2 𝑂: −2 (7 × −2) = −14
No charge on compound so equals 0: +2 − 14 + 2𝑥 = 0 = +6
Therefore 𝐶𝑟: +6
Redox Half Equations
• In a redox reaction, one substance always becomes reduced while the other one
oxidized
• To balance redox equations, we write out the half-equations for the oxidation and
reduction reactions
• Redox reactions can take place in neutral, acidic or basic solutions
o In acidic solutions, follow the steps below
o In neutral solution, balance the reaction as if it were in acidic solution
o In basic solution, instead of adding H+ ions add OH- ions
• In order to write a balanced redox reaction we must: write the two half equations first and
then add them together to get an overall equation (OHe)
TOPIC 9 REDOX PROCESSES NOTES
9.1 Oxidation and reduction
Oxidation and Reduction
o In oxidation and reduction reactions (also called redox reactions), electrons move
between atoms
o Oxidation involves the loss of Oxidation Reduction
electrons. The more a substance is
oxidized, the more positive it gets Loss of electrons Gain of electrons
o Reduction involves the gain of
electrons. The more a substance is Loss of hydrogen Gain of hydrogen
reduced the more negative it gets
o Remember: OIL RIG (Oxidation is Gain of hydrogen Loss of oxygen
loss, reduction is gain)
Oxidant and reductant
Definitions
Oxidant – A substance that readily oxidizes other substances
Reductant – A substance that readily reduces other substances
• An oxidizing agent makes oxidation
Oxidizing Reducing
happen. In order to make oxidation
agent/oxidant agent/reductant
happen, the agent must take the electron
from the substance
Gains electrons Loses electrons
• So, oxidizing agents are reduced
• Common oxidizing agents include Oxidizes another Reduces another reactant
halogens, ozone, maganate (VII) ions and reactant
hydrogen peroxide
• A reducing agent makes reduction Is reduced during the Is oxidized during the
happen. In order to make reduction reaction reaction
happen, the agent must give an electron
to the substance
• So, reducing agents are oxidized
• Common reducing agents include hydrogen, carbon, carbon monoxide and sulfur dioxide
, Oxidation state
• The oxidation state (also known as oxidation number) is the total number of electrons
that an atom either gains or loses to form a chemical bond with another atom – (the
charge an atom would have if the compound was composed of ions)
• Oxidation numbers are represented by a roman number. Example: Cu2O: Copper (I)
oxide, FeCl2: Iron (II) chloride
• Oxidation states can be used to identify which species have been oxidized and which
have been reduced
o An increase in oxidation number means oxidation (so reducing agent) (Loss
of electrons, becomes more positive)
o A decrease in oxidation number means reduction (so oxidizing agent) (Gain
of electrons, becomes more negative)
• Oxidation states are written as NUMBER and then SIGN
• To assign oxidation states to atoms in a molecule or compound we follow some very
specific rules:
Sum of oxidation state for a neutral Sum of oxidation state for a polyatomic ion: Ion
compound: 0 charge
Element by itself: 0 Group 1/2/3: Always +1/+2/+3
Monatomic ion: Ion charge D-Block elements have variable oxidation states
Halogens: Usually -1 (+1 with oxygen) Hydrogen: +1 (-1 with metals)
Oxygen: -2 (-1 in peroxide) Florine: Always -1
Question: Deduce the oxidation state of Cr in K2Cr2O7
𝐾: +1 (2 × +1) = +2 𝑂: −2 (7 × −2) = −14
No charge on compound so equals 0: +2 − 14 + 2𝑥 = 0 = +6
Therefore 𝐶𝑟: +6
Redox Half Equations
• In a redox reaction, one substance always becomes reduced while the other one
oxidized
• To balance redox equations, we write out the half-equations for the oxidation and
reduction reactions
• Redox reactions can take place in neutral, acidic or basic solutions
o In acidic solutions, follow the steps below
o In neutral solution, balance the reaction as if it were in acidic solution
o In basic solution, instead of adding H+ ions add OH- ions
• In order to write a balanced redox reaction we must: write the two half equations first and
then add them together to get an overall equation (OHe)