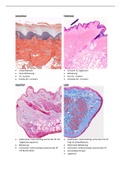
Any physical substance that occupies space and has mass:
solid, liquid, gas
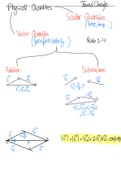
Mixtures Pure Substances
A substance made of at least Made of only 1 type of particle
two different types of pure → Separated into elements and compounds
substances mixed together.
Elements Compounds
A substance made up of 1 A substance made up of 2 or mo
Solutions: Mechanical type of atom elements BONDED together
Homogenous Mixtures: → ie. Periodic Table of
Mixtures Elements
Heterogenous
→ CANNOT see different
types of atoms/molecules
Mixtures
→ CAN see different
types of atoms/molecules
→ Example:
→ Example:
Atom The atom is m
Tiny particles that are the building blocks of 3 particles
of matter → Each element on the periodic
subatomic pa
table has a different type of atom.
Electrons, Pr
Oxygen Atom Neutro
Electrons Nucleus
Negatively charged The centre of the atom that includes 2
particles. subatomic particles: protons & neutrons
→ Almost no mass.
→ Orbits around the
nucleus
Protons Neutron
Positively charged Neutral particle
particles located in located in the
the nucleus of the nucleus.
atom. → Mass of
→ Mass of 1 1 atomic unit
atomic unit.