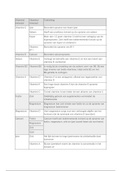
chemical bond ✔️the attractive force that holds atoms or ions together
ionic bonding ✔️a force that attracts electrons from one atom to another, which transforms a neutral
atom into an ion.
covalent bonding ✔️a bond formed when atoms share one or more pairs of electrons
nonpolar-covalent bond ✔️a covalent bond in which the bonding electrons are equally attracted to
both bonded atoms
polar ✔️describes a molecule in which the positive and negative charges are separated
polar-covalent bond ✔️a covalent bond in which a pair of electrons shared by two atoms is held more
closely by one atom
molecule ✔️neutral group of atoms held together by covalent bonds
molecular compound ✔️compound composed of molecules
chemical formula ✔️shows number of each kind of atom in a compound
molecular formula ✔️shows number of each kind of atom in a molecule4
bond energy ✔️the energy required to break the bonds in 1 mol of a chemical compound
electron-dot notation ✔️an electron configuration notation in which only the valence electrons of an
atom of the a particular element are shown, indicated by dots placed around the element's symbol