ATOMIC STRUCTURE
History of the development behind the nuclear model
Thomson Thomson discovered the existence of the electron in 1897 using cathode ray tubes .
> plum pudding model He is the scientist who proposed the idea of the plum pudding model of the atom The atomic .
> discovered electrons model at the time did not account for charged particles .
-
+
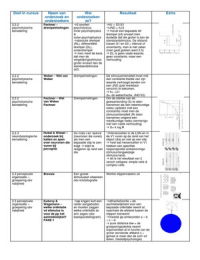
Thomson 's plum
pudding model consisted of a sphere made of protons -
-
l positively
charged soup ) embeded with negatively charged electrons . + -
+
1-
inbetweebiting posed massive nucleus
en
a
-
-
RuthFord 1911 , he discovered that there were 2 types of radiation ( alpha 1- beta particles) coming
the plum using the gold foil experiment
against from uranium
'
.
pudding model
/ Alpha particles were shot through a piece of gold foil -3 things
> atoms are mostly occurred to the alpha rays :
empty space and contain
'
most of the beams directly passed through the sheet , meaning
dense , positively that particles mostly consisted of empty space
charged nucleus some of the rays bounced back meaning that the center
'
.
( Of particles contained 1 the nucleus )
planetary model ) something dense
-
as alpha particles are positively charged , when hitting the nucleus it deflects due to
the protons it contains I like charges repel )
Bohr 1h 1912 Bohr joined Rutherford to create the finalised atomic model .
> significance & function His model shows the atom as a small , dense , positively charged nucleus surrounded by orbiting
of electrons electrons ( similar to the solar system) .
> final atomic model 1. Electrons that orbit the nucleus n :c 3. Radiation is emitted ( discharged )
<electrons exist on have a set size & energy .
n= ' when an electron changes to a
The of the orbit is lower energy orbit vice versa
principal energy levels 2.
energy ,
.
related to its size Ismail 4. Number Of electrons in the outer
orbit → low energy ) orbit determines the properties of
an element
, ATOMIC STRUCTURE
3 sub atomic particles
THE ATOM
-
:
neutrons protons electrons
11840 (5×10-4)
'
relative mass I 1 ->
in amu
definitions relative charge 0 + I -
I
nucleus nucleus levels
1- introductions position energy
mass number ctov isotopes ) :
proton -1 neutron ( nucleon number )
* neutrons help prevent atomic number 1 identifies the element ) : all isotopes of the same element will have the same
number of protons ( constant whole number )
protons from repelling ,
each other shorthand notations of atom / lions :
)
MAN NUMBER Charge Lions only
AFTER
smaller number is always the charge goes
at the bottom ! the number
ATOMIC NUMBER
relative atomic mall : the average mass of all the elements isotopes relative,
to 412
the mass of a carbon -12 atom .
how to calculate RAM - >
7 .
(add all males of isotopes by multiplying the ratio to mass) i. number of isotopes
2 if in percentage :
percentage ✗ mass number ( for all isotopes ) -
> add
together
(
.
37 100 ( divide by 100 )
35
>
eg . 1701 1701 (35×75)+(37×25)=35.5 (seen in the periodic table )
75-1 .
25-1 .
100
chemical properties remain the same as the number of electrons don't change
ISOTOPES } physical properties ( density , melting / boiling point , rate of diffusion ) changes
because the mass is different I > larger particle ,
slower movement
.
RADIO ISOTOPES - > an isotope of an element that has an unstable nucleus These . isotopes have an unstable
or radionuclide nucleus due to its excels nuclear energy The process of turning an unstable nuclei to .
a
( nuclear energy
stable nuclei is radioactive decay Radioactive decay emits gamma rays and / or
which
.
is held at the Nucleus subatomic particles , constitute ionizing radiation The .
energy can also be trans -
Ot an atom f- erred to an electron to release it at a conversion electron or to create I emit a particle
from the nucleus .
For chemical properties and sonnet of radiation ( tracers I. They are chemically
uses
their as
similar to non
-
radioactive atoms, therefore reaction sequences of certain atoms can be
mÉplo of traced using a radiation detector leg .
geiger muller counter ) .
( treatment )
' '
cells
revision guide They can also be used in
radiotherapy to kill malignant cancer .
firkin burns
radioactive contamination function of healthy
radiation poisoning damage tissues /
dAh GUVS org
-
, .
.
.
ans .
Exposure - > skin redness hair loss . . acute radiation syndrome c nausea ,
fatigue etc )
.
-
lead to mutation of cells ( cancer )
History of the development behind the nuclear model
Thomson Thomson discovered the existence of the electron in 1897 using cathode ray tubes .
> plum pudding model He is the scientist who proposed the idea of the plum pudding model of the atom The atomic .
> discovered electrons model at the time did not account for charged particles .
-
+
Thomson 's plum
pudding model consisted of a sphere made of protons -
-
l positively
charged soup ) embeded with negatively charged electrons . + -
+
1-
inbetweebiting posed massive nucleus
en
a
-
-
RuthFord 1911 , he discovered that there were 2 types of radiation ( alpha 1- beta particles) coming
the plum using the gold foil experiment
against from uranium
'
.
pudding model
/ Alpha particles were shot through a piece of gold foil -3 things
> atoms are mostly occurred to the alpha rays :
empty space and contain
'
most of the beams directly passed through the sheet , meaning
dense , positively that particles mostly consisted of empty space
charged nucleus some of the rays bounced back meaning that the center
'
.
( Of particles contained 1 the nucleus )
planetary model ) something dense
-
as alpha particles are positively charged , when hitting the nucleus it deflects due to
the protons it contains I like charges repel )
Bohr 1h 1912 Bohr joined Rutherford to create the finalised atomic model .
> significance & function His model shows the atom as a small , dense , positively charged nucleus surrounded by orbiting
of electrons electrons ( similar to the solar system) .
> final atomic model 1. Electrons that orbit the nucleus n :c 3. Radiation is emitted ( discharged )
<electrons exist on have a set size & energy .
n= ' when an electron changes to a
The of the orbit is lower energy orbit vice versa
principal energy levels 2.
energy ,
.
related to its size Ismail 4. Number Of electrons in the outer
orbit → low energy ) orbit determines the properties of
an element
, ATOMIC STRUCTURE
3 sub atomic particles
THE ATOM
-
:
neutrons protons electrons
11840 (5×10-4)
'
relative mass I 1 ->
in amu
definitions relative charge 0 + I -
I
nucleus nucleus levels
1- introductions position energy
mass number ctov isotopes ) :
proton -1 neutron ( nucleon number )
* neutrons help prevent atomic number 1 identifies the element ) : all isotopes of the same element will have the same
number of protons ( constant whole number )
protons from repelling ,
each other shorthand notations of atom / lions :
)
MAN NUMBER Charge Lions only
AFTER
smaller number is always the charge goes
at the bottom ! the number
ATOMIC NUMBER
relative atomic mall : the average mass of all the elements isotopes relative,
to 412
the mass of a carbon -12 atom .
how to calculate RAM - >
7 .
(add all males of isotopes by multiplying the ratio to mass) i. number of isotopes
2 if in percentage :
percentage ✗ mass number ( for all isotopes ) -
> add
together
(
.
37 100 ( divide by 100 )
35
>
eg . 1701 1701 (35×75)+(37×25)=35.5 (seen in the periodic table )
75-1 .
25-1 .
100
chemical properties remain the same as the number of electrons don't change
ISOTOPES } physical properties ( density , melting / boiling point , rate of diffusion ) changes
because the mass is different I > larger particle ,
slower movement
.
RADIO ISOTOPES - > an isotope of an element that has an unstable nucleus These . isotopes have an unstable
or radionuclide nucleus due to its excels nuclear energy The process of turning an unstable nuclei to .
a
( nuclear energy
stable nuclei is radioactive decay Radioactive decay emits gamma rays and / or
which
.
is held at the Nucleus subatomic particles , constitute ionizing radiation The .
energy can also be trans -
Ot an atom f- erred to an electron to release it at a conversion electron or to create I emit a particle
from the nucleus .
For chemical properties and sonnet of radiation ( tracers I. They are chemically
uses
their as
similar to non
-
radioactive atoms, therefore reaction sequences of certain atoms can be
mÉplo of traced using a radiation detector leg .
geiger muller counter ) .
( treatment )
' '
cells
revision guide They can also be used in
radiotherapy to kill malignant cancer .
firkin burns
radioactive contamination function of healthy
radiation poisoning damage tissues /
dAh GUVS org
-
, .
.
.
ans .
Exposure - > skin redness hair loss . . acute radiation syndrome c nausea ,
fatigue etc )
.
-
lead to mutation of cells ( cancer )